Press release -
New Study Sheds Light on Chemotherapy Resistance in Pancreatic Cancer Cells for Improved Therapeutics -- Kindai University
Osaka [9 Jun 2023] New Study Sheds Light on Chemotherapy Resistance in Pancreatic Cancer Cells for Improved Therapeutics
Researchers identify a key gene that gets silenced in chemotherapy-resistant pancreatic cancer and identify alternative druggable pathways
Advanced pancreatic cancer is typically treated with chemotherapeutic drugs like gemcitabine. However, resistance to these drugs over time may result in treatment failure. Researchers from Japan have now revealed the genetic mechanisms behind this drug resistance. Their findings show that resistant cells are deficient in the enzyme deoxycytidine kinase, and that they compensate by upregulating some metabolic pathways including nucleotide synthesis pathways. These can serve as alternative therapeutic targets for patients with gemcitabine-resistant pancreatic cancer.
Pancreatic ductal adenocarcinoma (PDAC), also known as ‘pancreatic cancer,’ is one of the most lethal cancers. With cases rapidly increasing around the world, it is predicted that PDAC may soon become the leading cause of cancer-related deaths. One of the main reasons PDAC is so lethal is that diagnosis usually occurs after the disease has advanced to the point where surgery is no longer an option. In such cases, chemotherapeutic drugs are often used to treat the disease. However, pancreatic cancer cells may develop resistance to these drugs, leading to treatment failure.
Gemcitabine is a first-line drug for PDAC with a structure similar to the nucleoside deoxycytidine. Unfortunately, the drug also faces the problem of resistance in cancer cells. To date, not much is known about the precise mechanisms that give rise to this resistance, and there has not been much success on overcoming it either.
In a new study published on May 1, 2023, in Molecular Cancer Research, scientists from Kindai University, Japan, made substantial progress towards understanding the nature of gemcitabine resistance in PDAC. Their findings provide valuable insights into how PDAC cells become resistant and what new druggable targets may arise as a result. “Although several combination therapies have been reported to sensitize gemcitabine-resistant PDAC cells, there is a need for further investigation of the mechanisms underlying chemoresistance to expand the therapeutic options,” explains Prof. Hitoshi Okada from the Department of Biochemistry, Faculty of Medicine, Kindai University, who led the study.
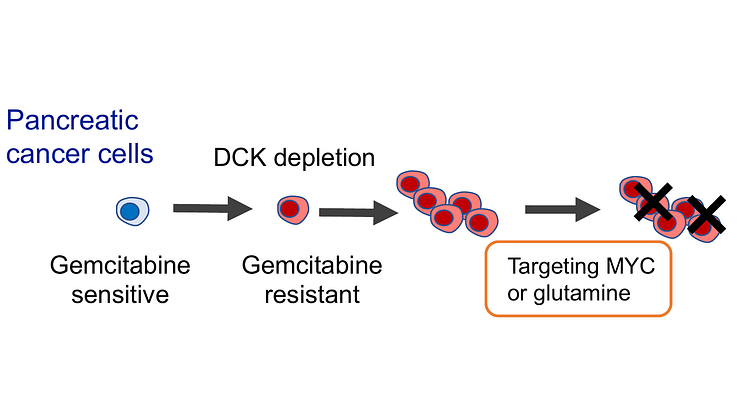
First, the researchers used human PDAC cell lines to screen for the genes most likely to be responsible for gemcitabine resistance. For this, they used the gene editing tool ‘CRISPR’ to create a knockout library that silenced the genes in pancreatic cell line populations. Upon treatment with gemcitabine, they observed which genes were inactivated most differently in the cells that survived treatment with gemcitabine when compared with the entire population before treatment. This led the team to a single gene that codes for a protein called deoxycytidine kinase (DCK), which took the center stage throughout the rest of their study.
Put simply, DCK is an enzyme involved in nucleotide metabolism; that is, it activates a pathway that provides cells with DNA precursors. The team wondered how the chemotherapy-resistant cancer cells managed to overcome the effects of lower levels of DCK, and whether these survival strategies could be potential therapeutic targets or "vulnerabilities." To this end, they conducted RNA-sequencing experiments in DCK-deficient PDAC cells to identify what other genes and pathways might be compensating for the lack of DCK.
These experiments revealed that DCK-deficient cells have an enriched expression of genes related to two key pathways: the MYC pathway and the glutamine metabolism pathway. Elaborating on this aspect, Suman Dash from Kindai University, the first author of the study, says, “Detailed molecular compensatory mechanisms of DCK deficiency are not entirely clear but the gene signatures we identified in DCK deficient cells are expected to be clinically important since the same gene signatures were observed in patients with low DCK expression.”
Taken together, the findings of this study not only explain the origin of gemcitabine resistance at the genetic level, but also shed light on how pancreatic cancer cells circumvent the side effects of acquiring this resistance. Armed with this new information, it may be possible in the future to find effective druggable targets in the pathways mentioned to combat resistant PDAC cells. “Our efforts provide insights into expanding the therapeutic approaches for this refractory disease,” concludes Prof. Okada, “Increasing the treatment options for chemotherapy-resistant pancreatic cancer will hopefully improve patient survival in the long term.”
Let us hope further research eventually leads us to a cure for this unruly disease!
Reference
Title of original paper: MYC/Glutamine Dependency Is a Therapeutic Vulnerability in Pancreatic Cancer with Deoxycytidine Kinase Inactivation-Induced Gemcitabine Resistance
Journal: Molecular Cancer Research
DOI: 10.1158/1541-7786.MCR-22-0554
URL https://doi.org/10.1158/1541-7786.MCR-22-0554
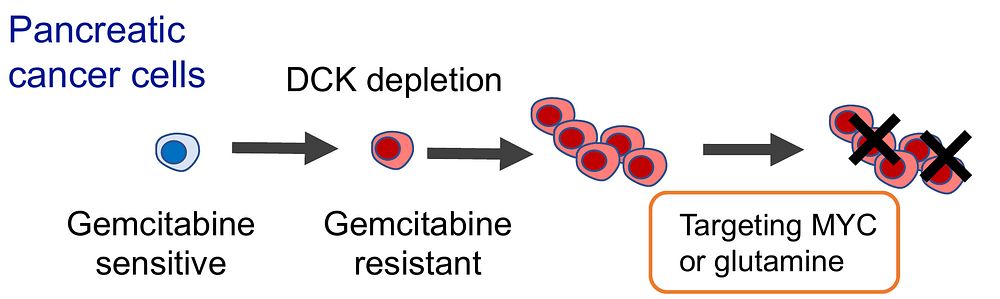
Image Title: Researchers elucidate the mechanisms behind gemcitabine resistance to improve treatment efficacy in pancreatic ductal adenocarcinoma
Image Caption: The study shows that pancreatic cancer cells that become resistant to gemcitabine do so by reducing their levels of an enzyme called deoxycytidine kinase and upregulating the MYC pathway and the glutamine metabolism pathway. Thus, targeting these pathways may be a viable therapeutic option for patients with resistant pancreatic cancer.
Image Credit: Hitoshi Okada from Kindai University
License type: Original content
About Kindai University
Kindai University was established in 1949 after the merger of Osaka Technical College (founded in 1925) and Osaka Science and Engineering University (founded in 1943). Over the past several decades, the university has transformed into a comprehensive educational organization with an ever-growing reputation. Kindai University has over 2,200 full-time faculty members, 6 campuses, and 18 research centers. As an academic institution offering a broad range of programs from across disciplines, Kindai University strives to impart practical education while nurturing intellectual and emotional capabilities. The university’s academic programs are fully accredited by Japan’s Ministry of Education, Culture, Sports, Science and Technology as well as by the National Institution for Academic Degrees and University Evaluation.
Website: https://www.kindai.ac.jp/english/
About Professor Hitoshi Okada from Kindai University
Dr. Hitoshi Okada is a Professor at Kindai University’s Department of Biochemistry/Graduate School of Medical Sciences. He primarily conducts research in the areas of metabolism, aging, epigenetics, DNA damage response, and tumor biology. He has several research papers to his credit. Professor Okada is a member of multiple prestigious association, including The Japanese Association for Molecular Target Therapy of Cancer, the American Association for Cancer Research, The Japanese Cancer Association, The Molecular Biology Society of Japan, and The Japanese Biochemical Society.
Funding information
This study was supported by the Japanese Government (MEXT) Scholarship program, a grant from Grant-in-Aid for Scientific Research on Innovative Areas, and the Vehicle Racing Commemorative Foundation.


