Press release -
Preclinical Stage Verification for Combined Use of a Drug Under Development with an Existing Drug to Stimulate the Immune System to Attack Cancer Cells - Kindai University
The research team from Kindai University’s Faculty of Medicine (Medical Oncology Division, Department of Internal Medicine), led by Assistant Prof. Koji Haratani, Lecturer Kimio Yonesaka and Senior Administrator Kazuhiko Nakagawa, in cooperation with DAIICHI SANKYO COMPANY, LIMITED (hereinafter “Daiichi Sankyo”), have verified at the preclinical stage that using the currently under-development anti-cancer drug, U3-1402※1, in combination with immune checkpoint inhibitors※2 (used to activate the immune system) has the potential to be an effective treatment.
U3-1402 is a type of “antibody-drug conjugate, ADC” with DNA topoisomerase I inhibitor, an antibody cytocidal that targets “HER3”※3 - a protein with a high level of expression in the cells of various solid cancers. The aim is to deliver cytocides with pinpoint accuracy, with safety and efficacy currently being verified with early-stage clinical trials.
In this preclinical trial, it was shown that U3-1402 attacked cancer cells, and at the same time as controlling cancer cell proliferation, due to the effect of the immune checkpoint inhibitors, immune cells adequately attacked cancer cells.
This study showed that this combined therapy is a prospect for future potential treatment strategies against a range of solid cancers.
The research paper was published online in the Journal of Clinical Investigation (impact factor: 12.282), a journal aimed at biomedicine.
1. Highlights of Research
- U3-1402 recognizes HER3, and efficiently delivers the cytocidal agent to the cancer cells.
- It is suggested that the tumor cell death caused by U3-1402 causes the release of an immuno-inducing substance, all types of immune cells are efficiently introduced to the site of the tumor and strengthen the effectiveness of the immune checkpoint inhibitors
- This is also promising as a possible treatment of cancer cells that have resistance to immune checkpoint inhibitors
2. Research Background
Cancer is broadly classified into two groups; hematologic cancer - cancer occurring in the lymph nodes and bone marrow - and solid cancers. PD-1/PD-L1 inhibitors are widely known immune checkpoint inhibitors that have exhibited effectiveness against a range of solid cancers, and are making extending the lives of many patients with advanced-stage cancers possible. PD-1/PD-L1 are proteins that are recognized by cancer and immune cells, and as they suppress immune cells within the body, attacks on cancer cells are prevented. However, PD-1/PD-L1 inhibitors interrupt this state of suppression, working to allow the immune cells to attack the cancer cells.
However, it is understood that the life-extending effects are experienced by only a portion of patients, and so the challenge of dealing with the cancer cells which are resistant to PD-1/PD-L1 inhibitors has been a pressing issue. In recent years, clinical trials have shown that combining PD-1/PD-L1 with cytocidal drugs has resulted in greater effectiveness of treatments, but the increased effectiveness could not be labelled as satisfactory; new strategies for developing treatments have been deemed necessary.
3. About Publication
Research paper title: U3-1402 sensitizes HER3-expressing tumors to PD-1 blockade by immune activation
Journal name: Journal of Clinical Investigation(impact factor: 12.282)
Co-authors: Koji Haratani (lead author), Kimio Yonesaka (corresponding author), Ryoji Kato, Naoki Mukawa, Naoto Kawakami, Kaoru Tanaka, Hidetoshi Hayashi, Masayuki Takeda, Kazuhiko Nakagawa - Internal Medicine (Medical Oncology Division) at the Faculty of Medicine, Kindai University.
Shiki Takamura, Masaaki Miyazawa - Department of Immunology, Faculty of Medicine, Kindai University
Osamu Maenishi - Department of Pathology, Faculty of Medicine, Kindai University
Naoyuki Maeda, Takashi Kagari, Kenji Hirotani - Daiichi Sankyo
Junji Tsurutani - School of Medicine, Showa University
Kazuto Nishio - Department of Genome Biology, Faculty of Medicine, Kindai University
Katsumi Doi - Department of Otorhinolaryngology, Faculty of Medicine, Kindai University
4. Details of Research
Commissioned by Daiichi Sankyo, this study made use of experimental animal models, cell cultures and clinical specimens to examine the effectiveness of the combined use of PD-1/PD-L1 inhibitors with U3-1402, a new anti-HER3 antibody-drug conjugate being clinically developed by Daiichi Sankyo, and the role played by U3-1402 in tumor immunity. U3-1402 acts specifically on HER-3 to efficiently and specifically damage HER3-expressing tumor cells. The tumor cells damaged by U3-1402 vigorously released HMGB-1, an immunostimulatory substance, and natural immune cells - such as dendritic cells, macrophages and NK cells - were introduced to the tumors’ locality. Further, it is believed that U3-1402 was demonstrated to improve localized antitumor ability and T-cell function, with the increased immune-stimulation due to the U3-1402 enhancing the therapeutic effectiveness of PD-1/PD-L1 inhibitors. In addition, these immunoreactive effects of U3-1402 and the combined effects with PD-1 inhibitors, were also observed in tumors with low sensitivity to PD-1 inhibitors - suggesting that U3-1402 may also be a key to overcoming PD-1/PD-L1 resistance. Moreover, using the clinical specimens (tumor tissue) of approx. 80 patients who had solid cancers that had received PD-1 inhibitor treatment, HER3 was immune-stained, with results showing that PD-1-resistant tumors frequently expressed HER3.
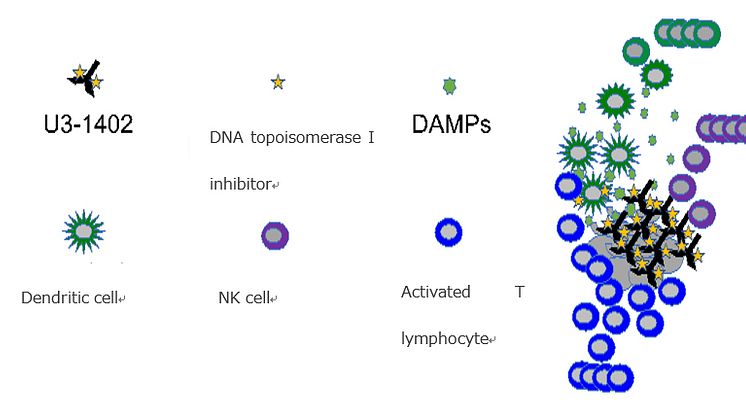
Explanation of diagram: U3-1402, which specifically recognizes the HER3 expressed in cancer cells, efficiently delivers DNA topoisomerase I inhibitor - a cytocidal - to the site of tumors. Due to the large amount of DNA topoisomerase I inhibitor※4 delivered to the cancer cells, the cells are powerfully damaged and release a large number of immune-stimulators called DAMPs (damage-associated molecular patterns). This induces accumulation of antigen-presenting cells, including dendritic cells, to the tumor site, and activates T lymphocytes. The activated T lymphocytes, along with NK cells that have also been aggregated by immune-stimulators, can additionally attack the cancer cells. The mechanism by which the use of PD-1/PD-L1 inhibitors in combined therapy further activate the functions of these immune cells was verified.
5. Future Outlook
U3-1402 is currently at the early clinical study stage for verification of safety and efficacy, and hereafter is anticipated to be further clinically developed. This is the first study to consider the combined efficacy of U3-1402 with immune checkpoint inhibitors, and because of this research further clinical development of combined therapy of U3-1402 with PD-1/PD-L1 is anticipated.
6. Glossary
*1 U3-1402
Antibody-drug conjugate currently being developed by Daiichi Sankyo. “DNA topoisomerase I inhibitor”, which inhibits the function of DNA topoisomerase - an enzyme that plays an important role in cell division, thus controlling the proliferation of cancer cells - is coupled to the antibody, anti-HER3, using a unique linker as an intermediary to form the antibody-drug conjugate.
*2 Immune checkpoint inhibitor
Immune checkpoint inhibitors strengthen immune cells’ attacks by inhibiting the brakes used for avoiding immune attacks.
*3 HER3
A protein expressed abundantly mainly in the surface of the cellular membrane of solid cancer cells. Although not necessarily expressed in all cancer cells, past research has shown a high frequency of expression in a large number of cancers.
*4 DNA topoisomerase I inhibitors
DNA topoisomerase I inhibitor is an enzyme necessary in the replication of DNA, by inhibiting this cancer cell proliferation can be blocked, and consequently cancer cells are killed.
[For inquiries regarding this study]
Kindai University, Faculty of Medicine / Hospital Office: Mr.Minamikawa, Mr.Ishii
Tel:+8172-366-0246 FAX:+8172-365-8300
E-mail:med.koho@itp.kindai.ac.jp
